 Nobel Prize winner for physics, Richard Feynman, comments that irreversibility is caused by the general accidents of life. When the gases mix, the most probable phenomenon occurs: degeneration into disorder. The most likely state is realized, namely the state of greatest entropy or disorder. The law ofincreasing entropy is therefore a law of probability, of statistical tendency toward disorder. In other words, such a hand is almost impossible a mixed hand, with a few cards of each suit, is the most probable. The probability of there being 13 hearts in a hand of bridge is 1 in 635 013 559 600. The disordered state occurred because it had the highest statistical probability. Entropy is a measure of the degree of disorder of the system. The 'orange' state is that of maximum disorder, the situation of greatest entropy because it was reached spontaneously from a situation of initial order. 
If they were originally mixed we would not expect to see them spontaneously separate into red and yellow. If we remove the wall we see that the two gases mix until there is a uniform distribution: an orange mixture. Suppose we have two gases, one red and one yellow, in two containers separated by a wall. In order to understand this better, it is useful to describe a model experiment: the mixing of gases. 'Entropy is in fact a measure of disorder and probability'. In this way the concepts of disorder and probability are linked in the concept of entropy. The direction is thus from order to disorder and entropy indicates this inexorable process, the process which has the maximum probability of occurring. Barry Commoner notes that sand castles (order) do not appear spontaneously but can only disappear (disorder) a wooden hut in time becomes a pile of beams and boards: the inverse process does not occur. 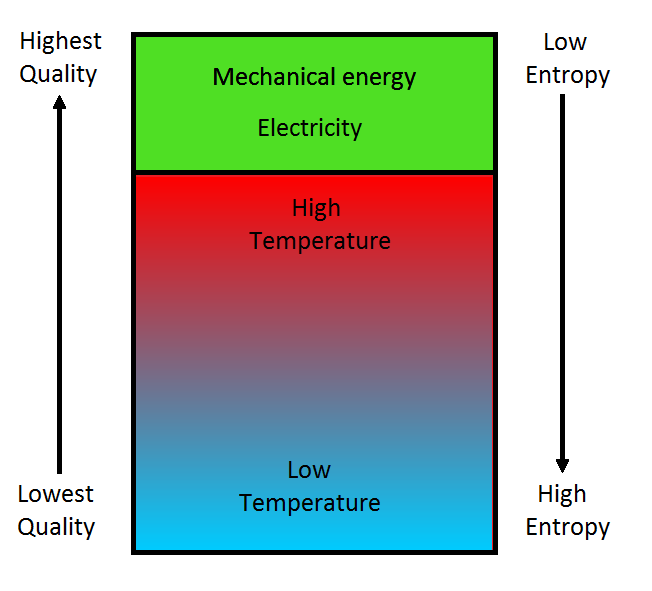
Maximum entropy, which corresponds to the state ofequilibrium ofa system, is a state in which the energy is completely degraded and can no longer produce work.Įntropy is therefore a concept that shows us the direction of events. 
With the concept of entropy Clausius (1865) reworded the second law of thermodynamics in a wider and more universal framework: Die Entropie der Welt strebt einem Maximum zu (The entropy of the world tends toward a maximum). The idea of the passage of time, of the direction of the transformation, is inherent to the concept of entropy. Transformations tend to occur spontaneously in the direction of increasing entropy or maximum dissipation. There is thus a spontaneous dissipation process which results in a degradation of energy toward lowest quality forms (heat). Perfume leaves a bottle and dissipates into the room we never see an empty bottle spontaneously fill. A jug that falls to the ground breaks (dissipation) into many pieces and the inverse process which could be seen running a film of the fall backwards never happens in nature. A ball bouncing tends to smaller and smaller bounces and dissipation of heat. The spontaneous tendency of energy to degrade and be dissipated in the environment is evident in the As stated before, the thermodynamic function known as 'entropy' (S) is a measure of the degree of energy dissipation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
